
Both bond length and bond energy changes as the bond order increases and as the number of electrons shared between two atoms in a molecule increases, the bond order of a bond increases, the strength of the bond increases and the distance between nuclei decreases (Table 9.10. In general chemistry courses, students learn that covalent bonds can come as either single, double or triple bonds, which are identifies by their bond order. To explain the observed paramagnetic properties of molecular oxygen with Molecular Orbital theory.From this diagram, calculate the bond order for O 2. You do not have the required permissions to view the files attached to this post. A video explaining the molecular orbital diagram and notation of C2- along with the bond order and magnetism. For C2, we need to consider the molecular orbitals formed by the combination of the atomic orbitals of two carbon atoms. Draw the molecular orbital diagram for the oxygen molecule, O 2. Molecular Orbital C2- Diagram, Bond Order, Magnetism. Example 6.9.2: M olecular Orbital Diagrams, Bond Order, and Number of Unpaired Electrons. 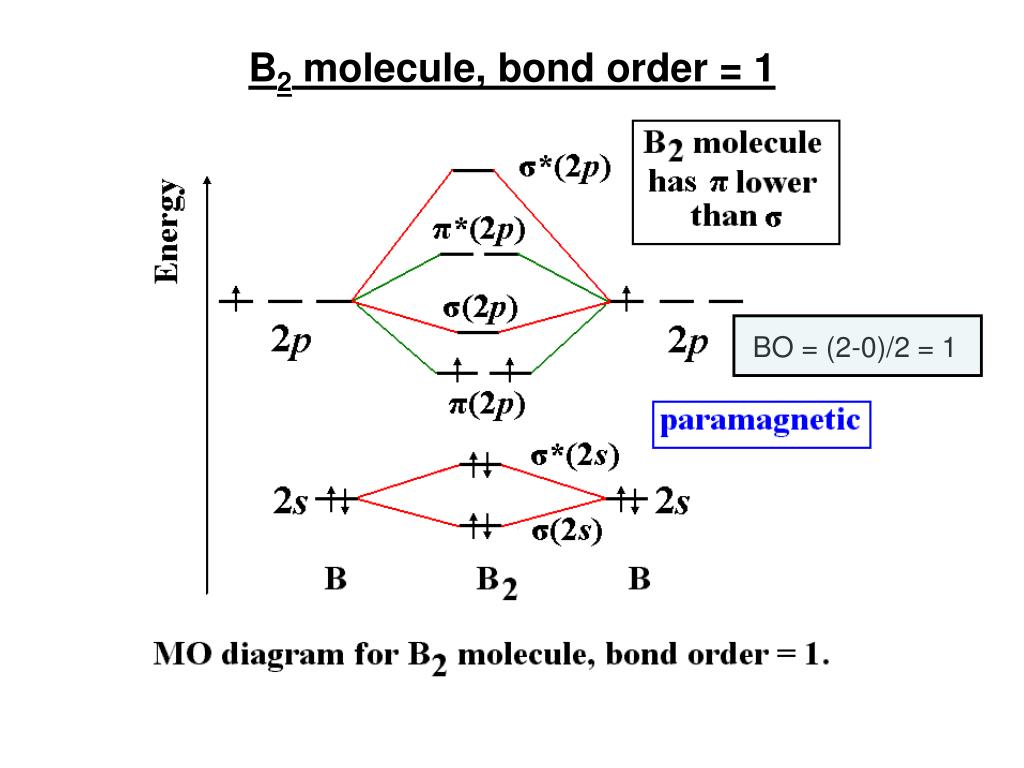
To describe the connection between bond order, bond length and bond energy in diatomic molecules As of December 2014, up to 46 of the energy in sunlight could be converted into electricity using solar cells.Like an atomic orbital, a molecular orbital is full when it contains two electrons with opposite spin.\) The bond order of the boron molecule is also calculated and. The region of space in which a valence electron in a molecule is likely to be found is called a molecular orbital ( Ψ 2). Draw the molecular orbital diagrams for C2-, C2, and C2+. This video discusses how to draw the molecular orbital (MO) diagram for the B2 (boron) molecule. Just like electrons around isolated atoms, electrons around atoms in molecules are limited to discrete (quantized) energies. One of the molecular orbitals in this molecule is constructed by adding the mathematical functions for the two 1 s atomic orbitals that come together to form this molecule. Using quantum mechanics, the behavior of an electron in a molecule is still described by a wave function, Ψ, analogous to the behavior in an atom. Molecular orbitals are obtained by combining the atomic orbitals on the atoms in the molecule. Molecular orbital theory describes the distribution of electrons in molecules in much the same way that the distribution of electrons in atoms is described using atomic orbitals. 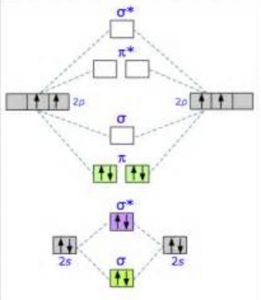
The 2s orbitals will overlap to form 2s and 2s. Each boron atom has one 2s and three 2p valence orbitals. We can ignore the 1s orbitals, because they do not contain the valence electrons. Then we rank them in order of increasing energy. Needs multiple structures to describe resonance Before we can draw a molecular orbital diagram for B, we must find the in-phase and out-of-phase overlap combinations for boron's atomic orbitals. 
Predicts the arrangement of electrons in molecules Predicts molecular shape based on the number of regions of electron density \): Comparison of Bonding Theories Valence Bond TheoryĬonsiders bonds as localized between one pair of atomsĬonsiders electrons delocalized throughout the entire moleculeĬreates bonds from overlap of atomic orbitals ( s, p, d…) and hybrid orbitals ( sp, sp 2, sp 3…)Ĭombines atomic orbitals to form molecular orbitals (σ, σ*, π, π*)Ĭreates bonding and antibonding interactions based on which orbitals are filled
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
